The discovery of membraneless organelles, their significance, and investigating an approach to study them systematically is the matter of contention for modern multidisciplinary research. Various membrane-less organelles exist as liquid droplets within the cell and arise from the condensation of cellular material in a process termed liquid-liquid phase separation and finely tunes many biochemical reactions.
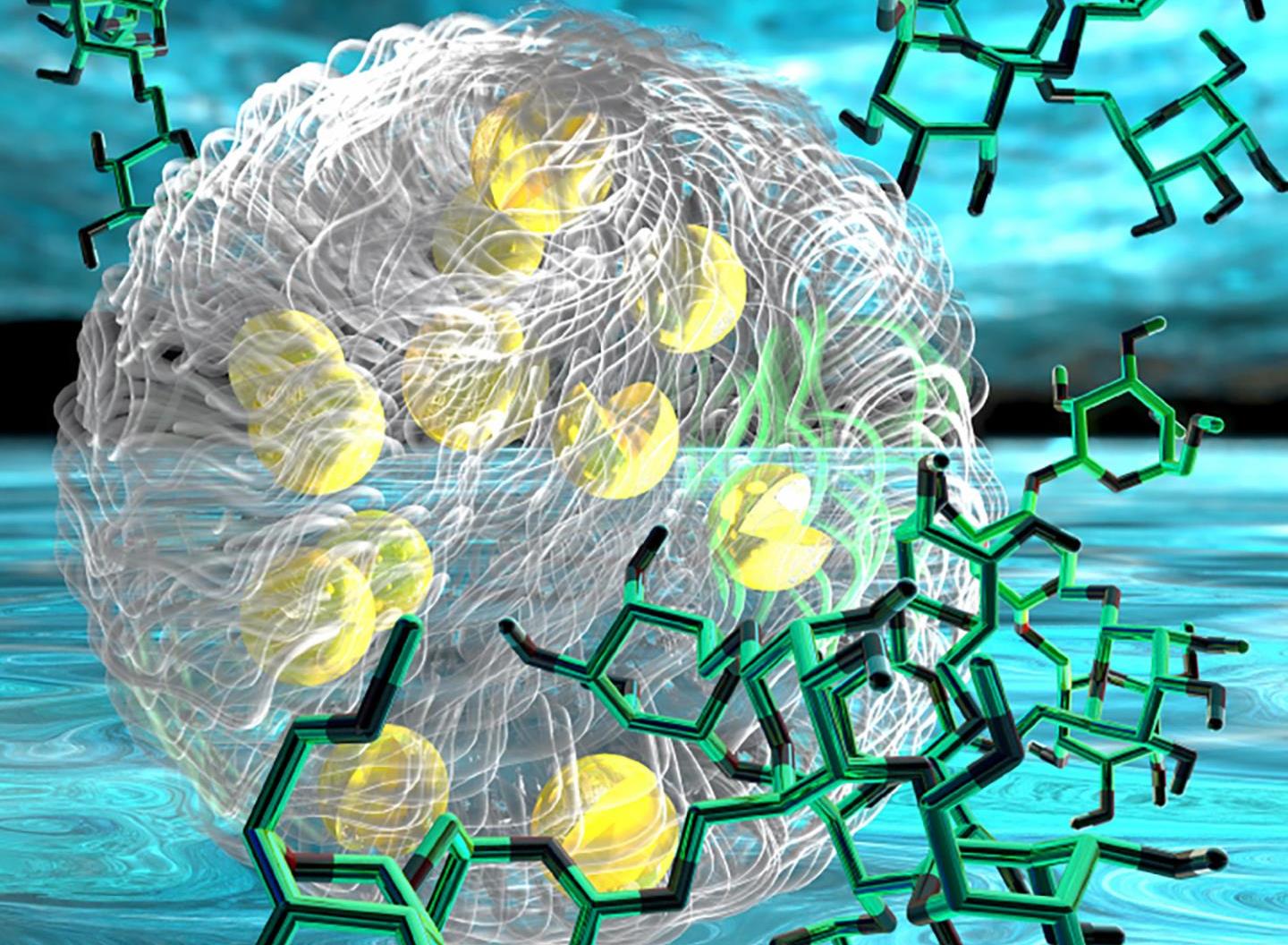
Researchers at Georgia Institute of Technology constructed a chemical blend in the laboratory which closely mimics membraneless organelles. This synthetic organelle is built out of some sugars, enzymes, a pinch of salt, a splash of polyethylene glycol, all carefully arranged in watery baths; in order to study some odd cellular biochemistry.
The outcomes of this research have been published in the journal ACS Applied Materials & Interfaces for the September 26, 2018 issue.
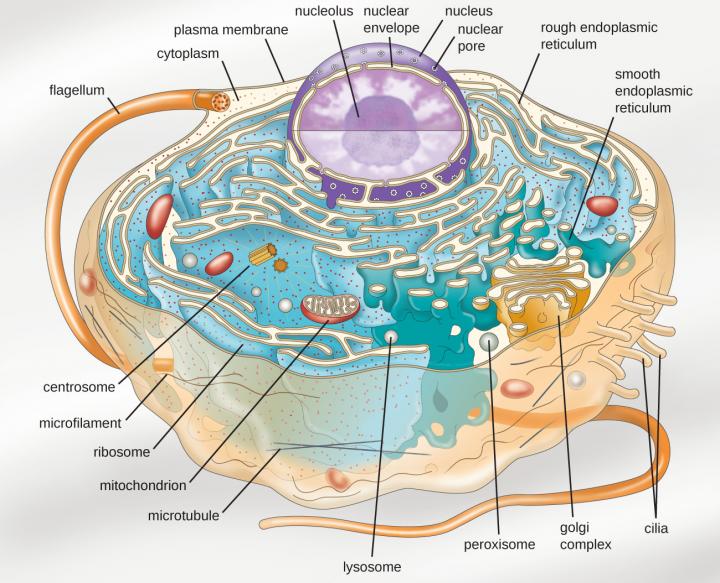
A prime example of membraneless organelle is nucleolus, which resides inside the nucleus. Earlier, scientists believed that during cell division nucleolus disappears and is then observed in the later stage. Meanwhile, the scientists found that nucleolus is devoid of the membrane and it gets diffused during cell division.
“After cell division, the nucleolus comes back together as a single compartment of fluid,” said Shuichi Takayama, the study’s principal investigator and a professor in the Wallace E. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University.
Membraneless organelles are made up of different aqueous solutions, each comprising of different solutes like proteins or sugar or RNA or salt. Contrasts in the thermodynamics of the arrangements of solutions, that is, the means by which their particles bob around, shield them from converging into a solitary arrangement.
“They’re all waters,” Takayama said. “They just don’t mix with each other because they have different solutes.”
Amid intermixing, critical things occur. The nucleolus, for instance, is fundamental to DNA transcription. The manufactured synthetic set-up, a gathering of watery arrangements made by the study’s first author, Taisuke Kojima, completed a less complex arrangement of responses that showed how the membraneless organelles could drive sugar processing.
“We had three phases of solutions that each held different reactants,” Kojima said. “It was like a ball with three layers: an outer solution, an intermediate solution, and a core solution. Glucose was in the outer layer; an enzyme, glucose oxidase, was in the second layer, and horseradish peroxidase was in the core along with a colorimetric substrate that gave us a visible signal when the last reaction we were looking for occurred.”
The glucose in the external layer interfaced with the glucose oxidase in the second layer, which catalyzed the glucose to hydrogen peroxide. It arrived in the second layer and interfaced with the horseradish peroxidase in the center layer, which catalyzed it in the center layer along with that compound that turns hues.
“This type of cascading reaction is what one would expect to see membraneless organelles perform,” Takayama said.
The cascade even transported every response item starting with one compartment then onto the next, something exceptionally run of the mill in natural procedures, similar to organs processing nourishment or an organelle preparing atoms. Some portion of the response shocked the researchers, and it brought about a novel disclosure.
“When researchers think about membraneless organelles, we often think that the reactions inside them are more efficient when their enzymes and substrates are in the same compartment,” Takayama said. “But in our experiments, that actually slowed the reaction down. We said, ‘Whoa, what’s going on here?'”
“When the substrate is in the same place where the product of the reaction also builds up, the enzyme sometimes gets confused, and that can impede the reaction,” said Kojima, who is a postdoctoral researcher in Takayama’s lab. “I was pretty surprised to see it.”
Kojima put the enzymes and substrate into discrete arrangements, which interfaced yet did not converge to a single solution, and the response in his synthetic organelle worked productively. This indicated how sudden nuances might be tweaking organelle science.
“It was a Goldilocks regime, not too much contact between substrate and enzyme, not too little, just right,” Takayama said. “Sometimes, in a cell, a substrate is not abundant and may need to be concentrated in its own little compartment and then brought into contact with the enzyme,” Takayama said. “By contrast, some substrates can be very abundant in the nucleus, and it might be important to partition them off from enzymes to get just enough contact for the right kind of reaction.”