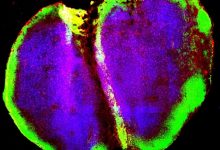
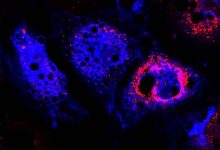
Signaling Pathway In Colon May Induce Inflammatory Bowel Disease
A new research led by Tokyo Medical and Dental University (TMDU) discovered the importance of cell signaling pathway in the growth of dysregulated immune cells which in turn induces inflammation in the colon.
Inflammation is localized biological reaction of the body tissue to harmful stimuli like damaged cells or irritants and is a protective response which involves immune cells, blood vessels, and molecular mediators. A new research led by Tokyo Medical and Dental University (TMDU) discovered the importance of cell signaling pathway in the growth of dysregulated immune cells which in turn induces inflammation in the colon.
As compared to other parts of the body, intestine comes across more foreign antigens and possible toxins, so it is not surprising that a tight regulation of immune for maintaining a healthy gut.This immune response protects the intestine against harmful pathogens but can endure harmless bacteria. However, an untimely immune response can induce chronic inflammation and the inflammatory bowel disease (IBD) can also be developed.
Macrophages are the ‘big eaters’ of the immune system which consume pathogens and hunt dying cells. Their precursors’ cells are called monocytes and they all are differentiated from the white blood cell. these together these differentiated cells altogether contribute to IBD pathology in the colon. Nevertheless, the essential mechanisms of this differentiation and how these macrophages obtain properties to induce inflammation of the colon were unclear.
In the laboratory, the team of Japanese researchers used a mouse model and induced IBD chemically and demonstrated that a protein signaling pathway is important for the generation of macrophages that cause colonic inflammation. The signaling pathway enhances access to the promoters of genes encoding inflammatory mediators, thus increasing their expression. The study results were recently published in Mucosal Immunology.
During their research, scientist found that there was increased expression of a protein that induces inflammatory responses from the Inflammatory monocytes and macrophages present in the colons of IBD mice compared with other cell populations in mice. They also expressed more transcription factor Stat1 that upregulates gene expression in response to stimulation by IFNγ, a signaling molecule released in the presence of pathogens.
“Mice lacking Stat1 expression differentiated fewer inflammatory macrophages from their monocyte lineage, and additionally developed less severe inflammation of their colons than control mice,” study first author Yusuke Nakanishi says. “This shows the importance of the IFNγ-Stat1 signaling pathway in colonic inflammation.”
Besides the fact that macrophages contributes to the differentiation of inflammatory monocytes and induces colonic inflammation, they also found that IFNγ signaling is important to maintain the macrophage population in inflamed colon.
“We already knew that the response to pathogenic bacteria in the gut involves remodeling of the DNA-protein package known as chromatin, enabling access for the expression of genes involved in inflammation,” the corresponding author Toshiaki Ohteki says. “This led us to investigate the role of IFNγ in a similar context, and we found that IFNγ loosened the chromatin structure around the promoters of two genes encoding inflammatory mediators, leading to an increase in their expression.”
This new understanding of how dysregulated macrophages develop in the colon during inflammation may lead to the identification of novel targets for IBD therapy.