A new study done by scientists at the Scripps Research Institute suggests that a genetic mutation which protects people from malaria and was assumed to be rare is unexpectedly common. This new study also led to the finding that humans who live in close quarters with malaria-carrying mosquitos may evolve defenses against the disease.
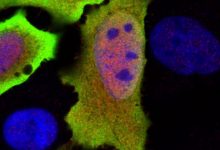
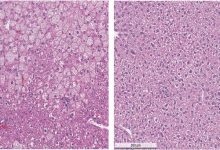
It has been demonstrated during the study that mutation in the gene PIEZO1 which translates into pressure-sensing protein, can lead to dehydration of red blood cells. During their study, they experimented in a mouse model and found that this mutation made it difficult for malaria causing parasite i.e. Plasmodium to infect red blood cells and cause cerebral malaria (a severe neurological complication of Plasmodium infection).
This condition of dehydrated red blood cell is called hereditary xerocytosis, was assumed to be very rare but the researchers were surprised to find it could be present in one in three people of African descent.“This syndrome is not rare anymore,” says Shang Ma, Ph.D., a research associate at TSRI.
According to the researchers, The mutation in PIEZO1 is not commonly found in non-African populations and had never been the focus of a large-scale analysis. The new findings suggest the mutation is much more common in areas where people have lived alongside selection pressure from malaria.
“This study is a good example of a host/pathogen arms race playing out in real-time—this time with the host a likely winner,” says Kristian Andersen, Ph.D., an assistant professor at TSRI and director of Infectious Disease Genomics at the Scripps Translational Science Institute (STSI).
Most of the previous studies take into account only the genetic condition called sickle cell disease, which makes it harder for Plasmodium to enter their red blood cells in People of African descent but this new study also linked PIEZO1 mutation with the malaria resistance. Going forward, Andersen says, large-scale genomic association studies will be needed to confirm the PIEZO1 mutation’s role in malaria resistance.
In the light of this study, the author Patapoutian has lab plans to study more about the biological role of PIEZO1 as well as to find how mutations in the protein could affect other health conditions. “The fact that we have a mouse model will make it seamless to test mechanisms behind any association we find in humans,” says Patapoutian. Indeed, PIEZO1 as a pressure sensor is important for cardiovascular development and function, and its deletion is proposed to cause hypertension.