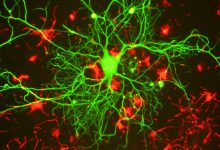
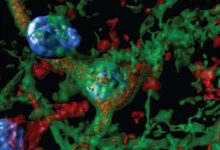
Scientists have discovered a connection between neuronal death and Lewy bodies which they believe is a key to treating Parkinson’s
Parkinson disease is a neurodegenerative disorder of central nervous system affecting nerve cells in the brain that produce dopamine. All over the world, around 10 million people are living with this disease which is incurable and leads to damage to motor control. There are two hallmarks which can be seen in the brain of the patient suffering from this disease which are dead brain cells and protein clumps. Dead brain cells are those producing dopamine while protein clumps are the Lewy bodies inside the neurons.
A new study led by Corinne Lasmézas, DVM, Ph.D., a professor on the Florida campus of The Scripps Research Institute (TSRI), discovered a connection between neuronal death and Lewy bodies which they believe is a key to treating Parkinson’s. The research, published recently in the journal Proceedings of the National Academy of Sciences, revealed the phenomenon through which neurons die off in the first place.
‘This study identifies the missing link between Lewy bodies and the type of damage that’s been observed in neurons affected by Parkinson’s,” says Lasmézas, senior author of the study. “Parkinson’s is a disorder of the mitochondria, and we discovered how Lewy bodies are releasing a partial break-down product that has a high tropism for the mitochondria and destroys their ability to produce energy.”
Lewy bodies inside neurons are formed from the clump of misfolded protein called α-synuclein (pα-syn*) whose function otherwise is related to the transmission of signals between neurons.
In the laboratory, Lasmézas and her team observed at cell cultures of neurons that were induced to accumulate fibrils made of misfolded α-synuclein, mimicking Lewy bodies in patients with Parkinson’s. they found that smaller protein clump is produced from the broken α-synuclein fibrils.
“Sometimes the nerve cells can efficiently degrade the α-synuclein fibrils, but if they get overwhelmed, the degradation may be incomplete,” she explains. “And it turns out that the result of that partial degradation, pα-syn*, is toxic.”
Furthermore, scientist marked the pα-syn* with an antibody so they could follow it throughout the cell after it was created. they found that pα-syn* traveled and attached itself to the mitochondria and after that mitochondrion starts breaking down which in turn leads to loss of ability of mitochondria to carry an electrochemical signal and produce energy. After carrying our same experiment in mouse and human brain samples, they confirmed the existence of pα-syn* in the dopamine-producing neurons.
“The Lewy bodies are big aggregates and they’re sitting in the cell, but they don’t come into direct contact with the mitochondria in the way pα-syn* does,” Lasmézas explains. “With Diego’s discovery, we’ve made a direct connection between the protein α-synuclein and the downstream effects that are observed when brain cells become damaged in Parkinson’s.”
In the light of this study, Lasmézas plans to continue learning more about the link between misfolded proteins and the destruction of mitochondria in neurons. “What we found may not be the only mechanism of toxicity, but we know it’s important,” she says. “This paper is about identifying where pα-syn* comes from and what it does to the mitochondria, but there’s obviously, mechanistically, a lot that we still don’t know.”
She says that these findings also have implications for designing treatments for Parkinson’s, noting that some drugs currently under development are focused on getting rid of larger fibrils that make up Lewy bodies.
“It’s important to be aware that when Lewy bodies are broken down, these toxic substances may be created,” Lasmézas says. In addition, she adds, the discovery of pα-syn* as an important component of the disease process points to a new target for creating drugs slowing disease progression.